




The everyday solution
for feeling like yourself
all month long
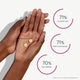
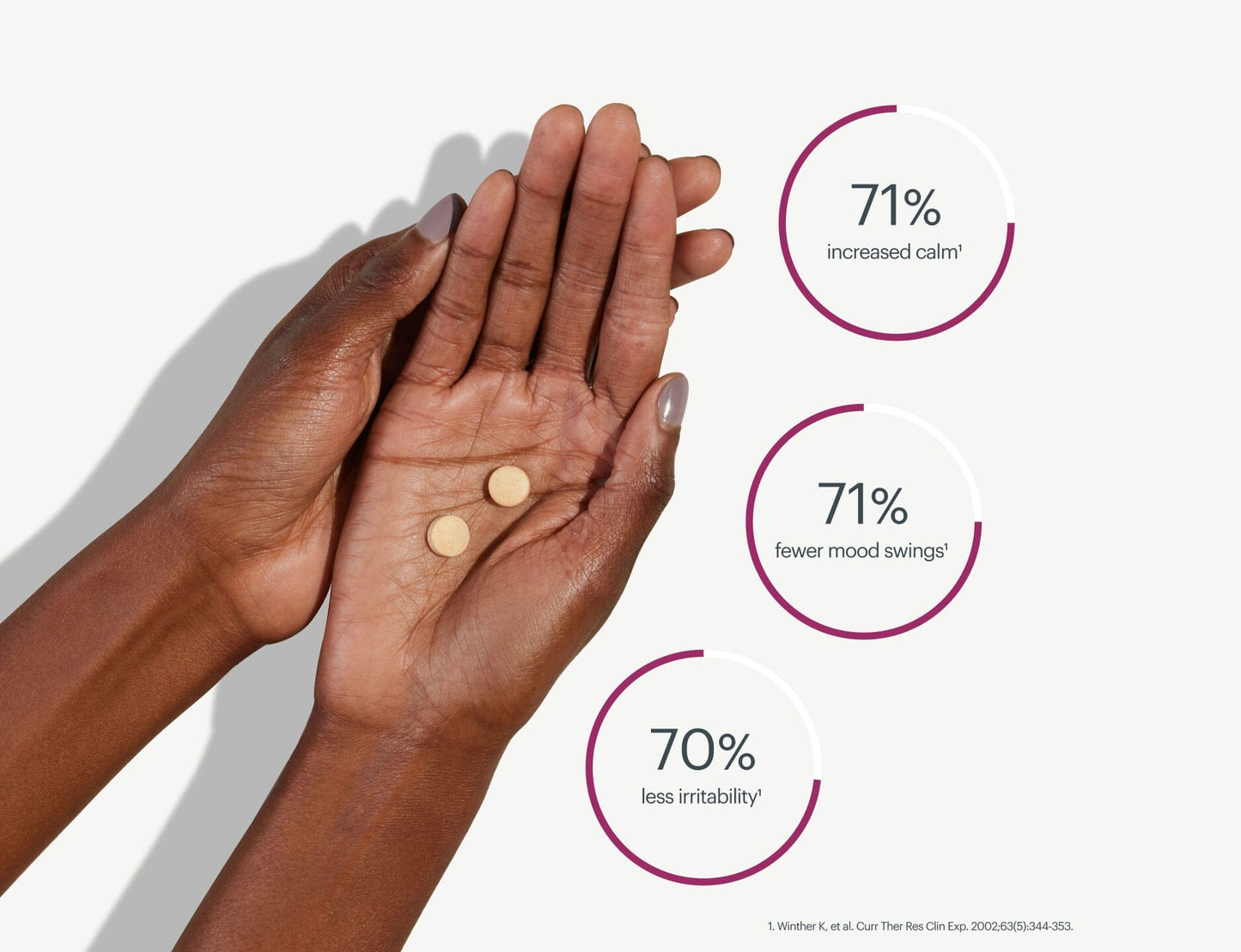
Calms irritability * ¹
Balances mood swings * ¹
Curbs cravings & reduces water retention * ²
Clinically tested, Scientifically validated
The proof is in the numbers. Bonafide rigorously researches and tests the safety and efficacy of our products.
77%
Women felt PMS relief within 3 months ³
60%
Women experienced reduced irritability and mood swings at month 3 ³
87%
Women have recommended Serenol to their friends ³

Add relief to your routine.
Mood swings are natural — but suffering is crazy

What to expect

We’ve unlocked the benefits of plant-based power.
Exclusive Bonafide Pollen
Exclusive Bonafide Pollen
Through our proprietary purification process, we unlock the unique healing benefits of pollen from four specific plants grown in Sweden. Exclusive Bonafide pollen reduces emotional symptoms like mood swings, irritability and uneasiness due to hormonal fluctuations like PMS and menopause.*
Royal Jelly
Royal Jelly
Food made for Queen bees, Royal Jelly is rich in nutrients, including vitamins and minerals, and has been shown with flower pollen to tackle irritability, uneasiness, and mood swings. *
Chromium Picolinate
Chromium Picolinate
Chromium Picolinate (CrPic) is an essential mineral for healthy metabolism, working hard to reduce calorie intake and fight off food cravings. *
Want to know more?
- What is Serenol?
- Serenol is a carefully formulated, hormone-free supplement for relieving emotional symptoms due to hormonal fluctuations like PMS and menopause, including irritability, mood fluctuations, and uneasiness.*
- How does Serenol work?
- The plant-based extract in Serenol is believed to work centrally in the body to reduce emotional symptoms due to hormonal fluctuations. Additionally, Serenol contains chromium picolinate, an essential mineral and insulin cofactor which improves glucose metabolism and appetite control.
- How long does Serenol take to work?
- Serenol takes time to work. In clinical studies, the majority of women experienced reduced irritability and mood swings results by month two, with improved results by month 3. While some women report feeling the benefits of Serenol earlier, we recommend you take Serenol every day for at least two months for best results.
- Does Serenol have any side effects?
- In clinical studies, any side effects reported were no different than those reported by people taking placebo tablets (sugar pills). If you have specific concerns, we recommend that you speak with your doctor.
- How do I know if Serenol is right for me?
- Serenol is intended for women who are experiencing emotional symptoms due to hormonal fluctuations like PMS and menopause, including irritability, mood fluctuations, and uneasiness.*
- Are Bonafide products covered by health insurance? By Medicare/Medicaid?
- At this time, our products are not covered by traditional insurance. That said, some healthcare HSA or FSA cards cover part or all of the cost of certain Bonafide products, so we recommend reaching out to your HSA/FSA provider to confirm if you can use your HSA or FSA funds.
Once you’ve confirmed that your HSA or FSA does cover Bonafide products, you should be able to place an order for any of our products; we’ve taken all of the necessary steps on our end to ensure you can access the proof of payment needed to file a reimbursement claim. We also recommend consulting your provider to determine what documentation is required to have your claim approved, as many providers require a Letter of Medical Necessity (LMN) in addition to a receipt. - What's the best way to place my FSA or HSA order?
- First, double-check the balance on your FSA or HSA account and that your plan covers Bonafide products. You can place an order online, using your FSA or HSA card (as you would any other credit card), or by calling our Customer Care Team for assistance at 1-833-266-2343, Monday to Friday 9AM - 6PM ET.
You’re not alone.
We’ve been there—really! Many of our support team members take our products, and all of us want to help you succeed. How can we help?



You deserve relief without compromise
That's why we've spent years rigorously researching, sourcing, and testing the purest ingredients to create safe and effective solutions.
Customer Reviews
Reviews represent individual experiences and may not reflect clinical results. Individual results may vary. Read here for our clinical results.